A new genome-wide CRISPR screening technique conducted by researchers at Vanderbilt University is offering new insights about how tumors in 80 to 90 percent of all cancers grow.


This novel approach developed by Maria Fomicheva, a graduate student in the lab of Ian Macara, Louise B. McGavock Professor and professor of cell and developmental biology, detected a genetic switch that induces continuous cell division, a marker of cancer initiation.
CRISPR, an acronym for “clustered regularly interspaced short palindromic repeats,” is one of the most significant scientific developments of the last decade. The technology allows researchers to edit or delete individual genes, numbering in the thousands per cell. A genome-wide CRISPR screen is like using Google within the human body to find singular genes responsible for specific traits. In their screen of epithelial cells—the barrier cells that cover skin and internal organs to protect the body from injuries and infections—Fomicheva and Macara selected for cells that have lost the ability to stop dividing when they reach a high density.
The researchers screened 40 million epithelial cells to avoid missing any potential hits, but finding a gene of interest among such a huge quantity is like finding a needle in a haystack. To solve this problem the authors developed a strategy to sort out cells using technology that marks different stages of the cell cycle with distinct colors. Their screen identified a well-known tumor suppressor called NF2, indicating that the researchers were on track.
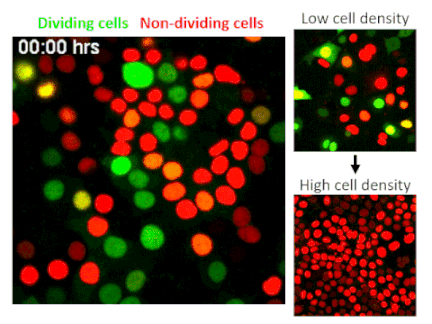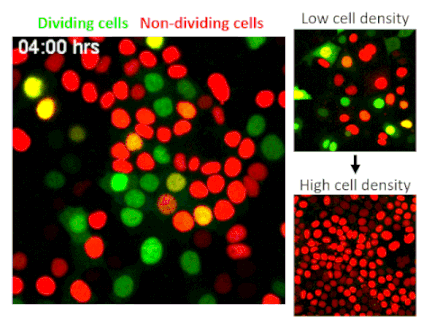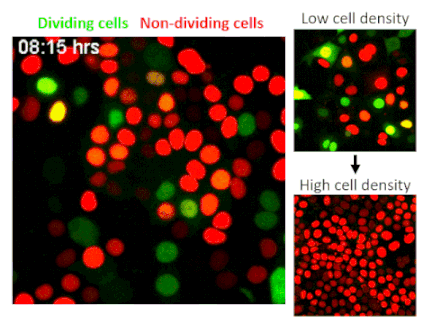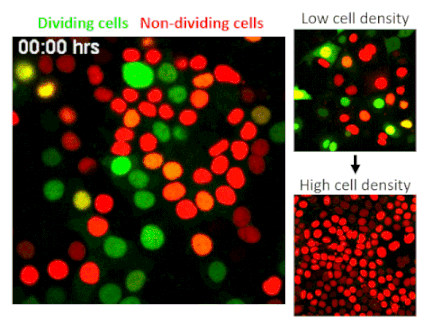
To their surprise, the researchers found that by deleting TRAF3, a protein that activates innate immunity, cells stop receiving a signal to rest. While TRAF3 has never previously been linked to density-dependent proliferation, this finding shows that without the protein, cells are triggered to continue dividing no matter how densely they grow. This trait is linked to cancer initiation, making its discovery potentially significant.
The article describing this research, “Genome-wide CRISPR screen identifies noncanonical NF-κB signaling as a regulator of density-dependent proliferation,” was published in the journal eLife on Nov. 13.
“Cell division is required for organ growth and self-renewal, but abnormal cell proliferation can cause organs to become misshapen or prevent them from functioning properly,” Fomicheva said. “It is known that normal, noncancerous cells decide when to stop dividing by sensing the cell density around them, but cancer cells lack this restraint and just keep dividing. It is important for us to understand the mechanisms that control this kind of cell behavior.”
Macara said that Fomicheva was brave to take on this project. “No one had ever performed a genome-wide screen in this way, and we were not sure if it would work or if we would find anything interesting,” he said. “Maria not only completed the entire screen on her own and discovered this surprising role for TRAF3, but went on to figure out the molecular mechanism by which it works.”
Fomicheva said further research is needed to determine what natural conditions lead to TRAF3 being disrupted, and whether these events play a role in leading to cancer.
The research was funded by the National Cancer Institute grant R35 CA132898. Vanderbilt resources that made this project possible were supported by the Vanderbilt-Ingram Cancer Center P30 CA68485 and the Vanderbilt Digestive Disease Research Center DK058404.