
Vanderbilt researchers have developed a new optical sensor that can accurately detect different types of inflammatory bowel disease and can be easily integrated into routine colonoscopy exams.
IBD is a group of inflammatory conditions of the colon and small intestine that affect more than one million Americans and can cause abdominal pain, vomiting, diarrhea, rectal bleeding, severe internal cramps, muscle spasms in the pelvic region and weight loss.
Current diagnostic and treatment procedures for IBD are inexact and rely largely on trial-and-error. In a paper published last month in the journal Biomedical Optics Express, the researchers report on a pilot study of a custom-developed endoscope that uses the chemical-fingerprinting technique Raman spectroscopy to detect molecular markers of IBD in the colon. It is the first sensor capable not only of objectively identifying the disease, but also of distinguishing between its two subtypes.
“With current methods, ultimately the diagnosis is dependent on how the patient responds to therapy over time, and you often don’t know the diagnosis until it’s been a few years,” said Anita Mahadevan-Jansen, the Orrin H. Ingram Professor Biomedical Engineering at Vanderbilt University, senior author of the paper, who initiated the project after a personal experience with IBD. “That’s why we decided to use a light-based method to probe the biochemistry of what’s going on in the colon. [rquote]Our goal is to use Raman spectroscopy to look at the actual inflammatory signals.”[/rquote]
Seeking a gold standard for diagnosis
IBD occurs in two subtypes: ulcerative colitis (UC), which is typically confined to the colon, and Crohn’s disease, which can occur in any part of the gastrointestinal tract. They can cause similar symptoms yet the two types respond differently to available treatments, which include drugs, dietary interventions and surgery.
Doctors currently use a combination of clinical, radiologic and pathological methods to diagnose a patient’s IBD subtype and guide treatment decisions, but these methods detect symptoms, rather than the underlying source of disease, and the process has lacked an objective gold standard. As a result, up to 15 percent of patients are diagnosed with indeterminate colitis, meaning the subtype is unknown, and an additional 5 to 14 percent have their IBD reclassified based on how they respond to treatment.
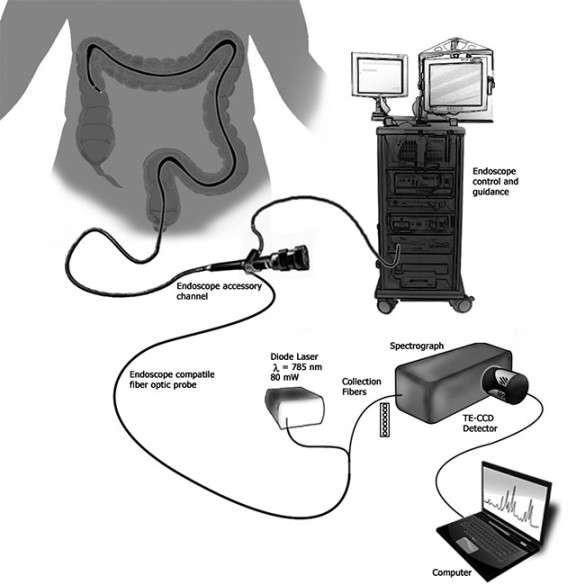
Creating a clinic-ready device
To develop the probe, the researchers first used tissue samples and animal models to identify preliminary biomarkers and spectral signatures consistent with IBD. Then they custom-built a portable Raman spectroscopy system capable of detecting these signatures.
“The customization was both the packaging – to make sure it can fit easily in the endoscopy channel and bend through the turns of the colon – and to probe the right depth in the tissues we are looking at to make sure we can get a robust measurement,” explained Mahadevan-Jansen.
Looking ahead
The researchers are now focused on refining a set of algorithms that help doctors interpret test results for a given patient. Ultimately, they envision a system that combines key information about the patient with Raman spectra to deliver, in a matter of seconds, tailored results that can inform a diagnosis or help track a patient’s response to treatment.
“It’s a unique way of thinking about personalized medicine that takes into account all sorts of information – beyond just the genome – including demographics and many other factors that make a person unique,” said Mahadevan-Jansen. “Our lab has really pushed to find ways to apply optical technology to support this kind of personalized medicine.”
The researchers are also pursuing studies in animal models to further understand the sources of various molecular signals, identify the types of changes that can be expected as a result of treatment and determine whether the device could be used to detect IBD or risk factors at even earlier stages.
Optical Society of America News Release: New Sensor Integrates Inflammatory Bowel Disease Detection into Colonoscopy Procedure