It’s now official. Tennessee has joined California to become only the second American state memorialized in the periodic table of elements.
The International Union of Pure and Applied Chemistry (IUPAC), the scientific body that validates the discovery of new elements and approves their official names, just gave its final approval of the name ‘tennessine’ for the newly discovered superheavy element 117.
Californium, element 98, was discovered in the 1950s and named for the university and state of California.
“It’s a great day for the state of Tennessee, and we’re grateful that our research has helped make this recognition possible,” said Joseph Hamilton, Landon C. Garland Distinguished Professor of Physics at Vanderbilt University, who played a pivotal role in the discovery. “We’ll be in the textbooks forever.”

The international collaboration that discovered the new element included Hamilton and Vanderbilt Professor of Physics A.V. Ramayya, along with scientists from Oak Ridge National Laboratory, the University of Tennessee-Knoxville, the Joint Institute for Nuclear Research in Russia and Lawrence Livermore National Laboratory in California. Hamilton initially proposed the name tennessine to honor the state where three of the participants – Vanderbilt, Oak Ridge and the University of Tennessee – are located. The name was agreed upon by the entire team, which submitted it to IUPAC.
The approval process began in December 2015 when IUPAC and the International Union of Pure and Applied Physics announced that they had verified the existence of superheavy element 117, more than five years after the scientists had first reported its discovery.
The name is spelled tennessine instead of tennessium because the new element is classified as a halogen-like chlorine and fluorine. Halogens are given names that end in the suffix “-ine.” Tennessine’s symbol in the periodic table is Ts.
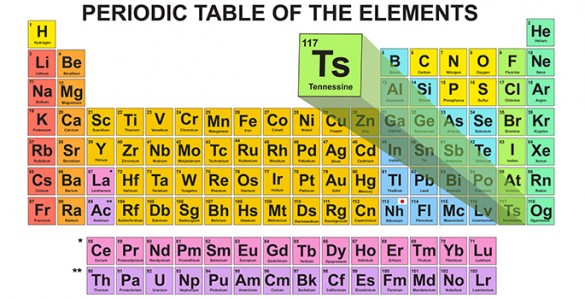
The discovery of tennessine has an importance beyond helping to complete the seventh row of the periodic table. According to Hamilton, it provides evidence for the existence of the long-sought, theoretically predicted “island of stability.” The concept of an “island of stability” is that there is a group of superheavy elements that should have increased stability and novel physical and chemical properties that could lead to new technologies, such as compact energy sources, if they can be created.
Related material:
Tennessee may become second state in periodic table
Vanderbilt physicist plays pivotal role in discovery of new super-heavy element