
The circadian clocks that control and influence dozens of basic biological processes have an unexpected “snooze button” that helps cells adapt to changes in their environment.
A study by Vanderbilt University researchers published online Feb. 17 by the journal Nature provides compelling new evidence that at least some species can alter the way that their biological clocks function by using different “synonyms” that exist in the genetic code.
“This provides organisms with a novel and previously unappreciated mechanism for responding to changes in their environment,” said Professor of Biological Sciences Carl Johnson. He and Associate Professor of Biological Sciences Antonis Rokas collaborated on the study.
Like many written languages, the genetic code is filled with synonyms: differently spelled “words” that have the same or very similar meanings. For a long time, biologists thought that these synonyms, called synonymous codons, were in fact interchangeable. Recently, they have realized that this is not the case and that differences in synonymous codon usage have a significant impact on cellular processes, so scientists have advanced a wide variety of ideas about the role that these variations play.
The new insight is not only an important advance in understanding evolution at the molecular level, but it also has potential applications in biotechnology, such as biofuel production, and gene therapy.
“While biological clocks are vital to maintaining healthy patterns of sleep, metabolism, physiology and behavior, under certain environmental conditions strict adherence to these rhythms can be disadvantageous,” said Michael Sesma of the National Institute of General Medical Sciences, which partially funded the work. “[rquote]This work shows how organisms can ignore the clock under certain circumstances—much like hitting a biological snooze button on the internal timepiece—and enhance their survival in the face of ever-changing circumstances.”[/rquote]
The basic letters of the genetic code are a quartet of molecules (nucleic acids) designated A, C, G and U. These are combined into 61 triplets called codons, which are analogous to words. The codons provide the blueprints that the cell’s protein-building machinery uses to generate amino acids, which are the basic building blocks that make all the proteins found in living organisms. However, cells only use 20 amino acids. That means a number of amino acids are produced by several different codons. For example, CCA, CCG and CCC are synonymous codons because they all encode for the same amino acid, proline.
It turns out that there is a reason for this redundancy. Some codons are faster and easier for cells to process and assemble into proteins than others. Recognition of this difference led to the concept of optimal codons and the hypothesis that natural selection should drive organisms – particularly fast growing ones – to use genes that use optimal codons to make critical proteins that need to be highly abundant or synthesized rapidly in cells.
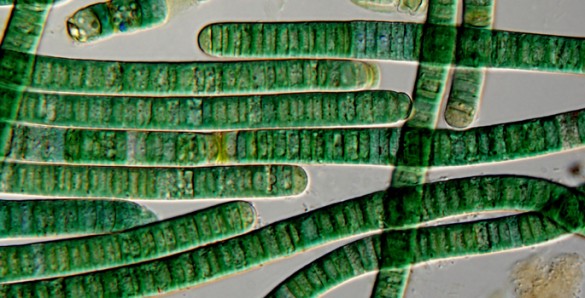
The problem with this hypothesis was shown by Johnson and Rokas’ study of the effect of changing codon usage on the simple biological clock found in single-celled cyanobacteria (blue-green algae) and a similar study of the more complex biological clock found in bread mold performed by a team led by Professor Yi Liu of University of Texas Southwestern Medical Center that were published together.
“What the Liu team found was that optimizing all the codons used by the fungal biological clock knocked the clock out, which was totally unexpected! Those researchers concluded that clock proteins in the fungus are not properly assembled if they are synthesized too rapidly; it’s as if the speed of one’s writing affected our ability to read the text,” Johnson summarized.
In the cyanobacteria, however, the researchers observed a different phenomenon. At Vanderbilt, Research Associate Professor Yao Xu optimized the codons in the cyanobacteria’s biological clock. This did not shut the clock down in the algae, but it did have a more subtle, but potentially as profound effect: It significantly reduced cell survival at certain temperatures.
“Xu figured that the biological clock with optimized codons might work better at lower temperatures and it did,” Johnson said. However the substitution also modified the biological clock so it ran with a longer, 30-hour period. When forced to operate in a 24-hour daily light/dark cycles, the bacteria with the optimized clock grew significantly slower than “wild-type” cells. “In cyanobacteria, it’s as if writing speed changes the meaning,” said Rokas.
The potential importance of changes in synonymous codon usage in adapting to environmental factors is magnified by the fact that they can influence the operation of biological clocks, which function as a key adaptation to daily environmental rhythms. Biological clocks control and influence dozens of different basic biological processes, including sleeping and feeding patterns, core body temperature, brain activity, hormone production and cell regeneration.
“It is now clear that variations in codon usage is a fundamental and underappreciated form of gene regulation,” said Rokas.

Recognition of the importance of this process has a number of potential applications in biotechnology. For example, “it should be possible to improve the ability of algae to robustly express biofuel-producing proteins from other organisms by optimizing the codons that they use,” Johnson said.
Vanderbilt graduate student Peijun Ma, postdoctoral fellow Premal Shah from the University of Pennsylvania and Yi Liu, professor at the University of Texas Southwestern Medical Center also contributed to the study, which was funded by grants from the National Institute of General Medical Sciences (GM067152, GM088595, GM068496 & GM062591), the Welch Foundation (I-1560), the National Science Foundation (DEB-0844968), the Burroughs Wellcome Fund and a David and Lucille Packard Foundation Fellowship.