by Kelsey Herbers
Vanderbilt University Medical Center is participating in an international study to determine the effectiveness of a new therapeutic vaccine for treating women with precancerous changes on the cervix.
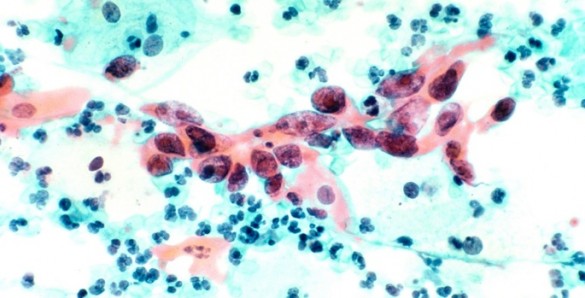
These precancerous lesions, also called dysplasia or cervical intraepithelial neoplasia (CIN), are caused by an infection of the human papillomavirus (HPV). The abnormalities are asymptomatic but can be detected by standard Pap test screening and diagnosed by a colposcopy and biopsy of the affected area.
The severity of CIN is typically categorized into three grades. CIN1 — which is often cleared by a woman’s own immune system without any treatment — is considered low grade, while CIN2 and 3 are referred to as high grade. Untreated, these high-grade abnormalities could progress to cancer over time, so surgical excision of this area of the cervix is usually recommended.
The study, sponsored by Inovio Pharmaceuticals, treats women with high-grade CIN with a new investigational therapeutic vaccine that targets HPV at the source of the cervical abnormalities. The goal is to show the vaccine is a safe and effective alternative to surgery, which can weaken the cervix, putting patients at risk of premature delivery with future pregnancies.
Three doses are given — one on the day of enrollment, one four weeks into participation and another three months following enrollment. If effective, the vaccine will cause the CIN lesions to regress.
Participating patients will be carefully monitored for a total of two years to evaluate regression of the lesion and to ensure the cervical abnormality doesn’t return.
Because the development of cervical cancer is slow — meaning the progression from the first HPV infection of the cervix through the three grades of CIN and potentially onto invasive cancer usually takes five to 10 years — there is minimal risk associated with delaying surgical treatment while the effectiveness of the vaccine is evaluated.
“My hope is that women with CIN2 or 3, especially those considering future pregnancies, will consider this option to surgical treatment,” said Howard Jones III, MD, professor of Obstetrics and Gynecology and a leader of Vanderbilt’s study. “We would love to avoid surgical treatment in all women if we could, especially in younger women who haven’t yet had their families.”
Providers who have patients with high-grade squamous intraepithelial lesion (HSIL) as determined through their Pap test can refer patients for participation in the study by contacting Jones at 615-322-8072.