The epithelial cells that line the intestines fire bacteria-fighting “bullets” into the gut, Vanderbilt University researchers have discovered.
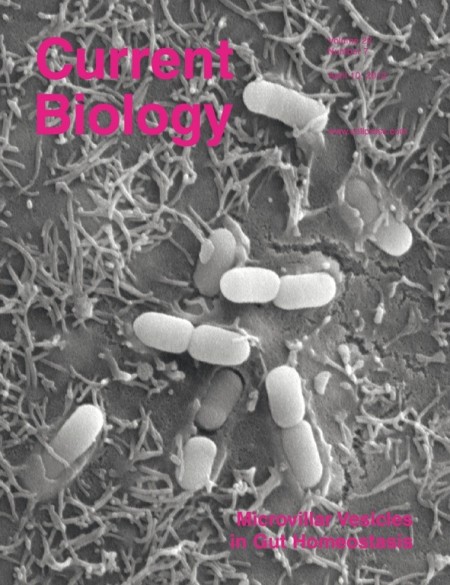
The findings, featured on the cover of the April 10 issue of Current Biology, represent a new mechanism for defending the body against gut microbes.
The surfaces of intestinal epithelial cells are covered by microvilli – specialized structures that project, like thousands of tiny fingers, into the lumen of the gut. The “textbook” view of the microvilli is that they increase the cellular surface area for absorbing and processing nutrients, said Matthew Tyska, associate professor of cell and developmental biology.
“Our studies suggest a new function for the microvilli in regulating epithelial-microbial interactions,” he said.

Tyska and his colleagues had previously demonstrated that the microvilli launch vesicles – tiny bubble-like packets of cell membrane – into the lumen, and that these vesicles contain high levels of an enzyme called intestinal alkaline phosphatase (IAP). Because IAP reduces the toxicity of bacterial products and limits their pro-inflammatory signaling, the investigators suspected that the vesicles might protect against bacteria and other pro-inflammatory compounds.
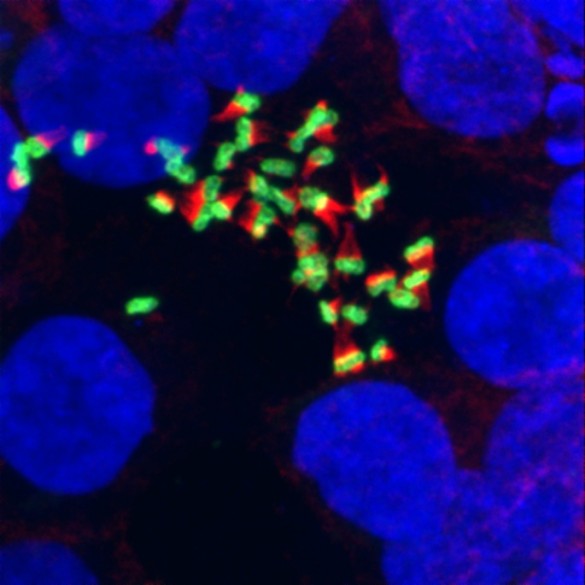
They show in the current studies that IAP in vesicles isolated from rat intestines is able to detoxify bacterial products from multiple types of bacteria. Using electron microscopy, they also observed that the vesicles “pile up” on the surface of bacteria, which may facilitate their clearance from the gut.
To study how the vesicles affect bacterial function, they incubated cultures of human intestinal epithelial cells with enteropathogenic E. coli (EPEC) in the presence or absence of vesicles. EPEC is a type of “adherent pathogenic bacteria” that gets introduced to the lumen when someone eats contaminated meat, for example, Tyska said. EPEC tries to bind to the microvilli, inject its products into the cell and destroy the brush border (the layer of densely packed microvilli).
“Bugs that destroy the brush border are bad news; you cannot survive without microvilli,” Tyska said. “EPEC is a major human health problem, especially in developing countries.”
To the researchers’ surprise, the vesicles were able to prevent EPEC from binding to and destroying the microvilli.
“Anything that can keep adherent pathogenic bacteria off of host cells and in the lumen is pretty exciting,” Tyska said.
Not only did the vesicles act as decoys for EPEC binding, they inhibited bacterial growth and even killed bacterial cells, the investigators found. In addition, adding EPEC to the cultured cells stimulated expression of IAP and vesicle production.
“Our findings are consistent with the production of these vesicles being a regulated host defense process,” Tyska said. “When cells sense the accumulation of bacteria or bacterial toxins in their environment, they respond by cranking up alkaline phosphatase levels and vesicle production.
“Because trillions of microvilli extend into the lumen, this is going to be a very effective way of quickly conditioning the lumen against bacterial pathogens,” he said.
The investigators are currently studying the process in mice that have deficiencies in vesicle production. They expect to see chronic inflammation, changes in the microbiome (the bacterial species that normally inhabit the gut), and “other changes that push them away from normal physiology,” Tyska said. They also plan to use live tissue imaging to follow where the vesicles go in the gut lumen.
The research, which began with Tyska’s interest in a motor protein that is enriched in microvilli, “is, I think, a great example of how doing fundamental research on molecules can lead to new insights about physiology and pathophysiology,” he said.
Contributors to the current studies include first author David Shifrin, Russell McConnell, Ph.D., Rajalakshmi Nambiar, Ph.D., James Higginbotham, Ph.D., and Robert Coffey, M.D. The research was supported by National Institutes of Health grants from the National Institute of Diabetes and Digestive and Kidney Diseases and the National Cancer Institute, by grants from the American Heart Association and by a Vanderbilt University Innovation and Discovery in Engineering and Science Award.












